MYB-QKI Rearrangements in Angiocentric Glioma Drive Tumorigenicity Through a Tripartite Mechanism
Abstract
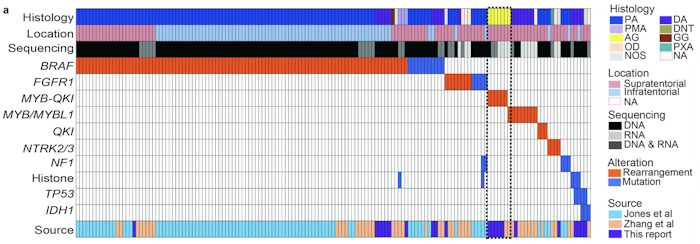
Angiocentric gliomas are pediatric low-grade gliomas (PLGGs) without known recurrent genetic drivers. We performed genomic analysis of new and published data from 249 PLGGs, including 19 angiocentric gliomas. We identified MYB-QKI fusions as a specific and single candidate driver event in angiocentric gliomas. In vitro and in vivo functional studies show that MYB-QKI rearrangements promote tumorigenesis through three mechanisms: MYB activation by truncation, enhancer translocation driving aberrant MYB-QKI expression and hemizygous loss of the tumor suppressor QKI. To our knowledge, this represents the first example of a single driver rearrangement simultaneously transforming cells via three genetic and epigenetic mechanisms in a tumor.