Overcoming resistance to single-agent therapy for oncogenic BRAF gene fusions via combinatorial targeting of MAPK and PI3K/mTOR signaling pathways
Abstract
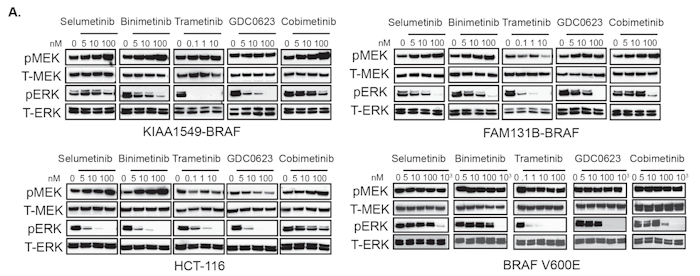
Pediatric low-grade gliomas (PLGGs) are frequently associated with activating BRAF gene fusions, such as KIAA1549-BRAF, that aberrantly drive the mitogen activated protein kinase (MAPK) pathway. Although RAF inhibitors (RAFi) have been proven effective in BRAF-V600E mutant tumors, we have previously shown how the KIAA1549-BRAF fusion can be paradoxically activated by RAFi. While newer classes of RAFi, such as PLX8394, have now been shown to inhibit MAPK activation by KIAA1549-BRAF, we sought to identify alternative MAPK pathway targeting strategies using clinically relevant MEK inhibitors (MEKi), along with potential escape mechanisms of acquired resistance to single-agent MAPK pathway therapies. We demonstrate effectiveness of multiple MEKi against diverse BRAF-fusions with novel N-terminal partners, with trametinib being the most potent. However, resistance to MEKi or PLX8394 develops via increased RTK expression causing activation of PI3K/mTOR pathway in BRAF-fusion expressing resistant clones. To circumvent acquired resistance, we show potency of combinatorial targeting with trametinib and everolimus, an mTOR inhibitor (mTORi) against multiple BRAF-fusions. While single-agent mTORi and MEKi PLGG clinical trials are underway, our study provides preclinical rationales for using MEKi and mTORi combinatorial therapy to stave off or prevent emergent drug-resistance in BRAF-fusion driven PLGGs.