Tracing Evolution History of 100 Whole Genome Sequences of Diffuse Stem Brain Tumor
Abstract
Diffuse intrinsic pontine glioma (DIPG) is a deadly disease among young children. The evolution path and
mutational processes giving rise to DIPG remain elusive. We analyzed 100 whole genome sequences (WGS)
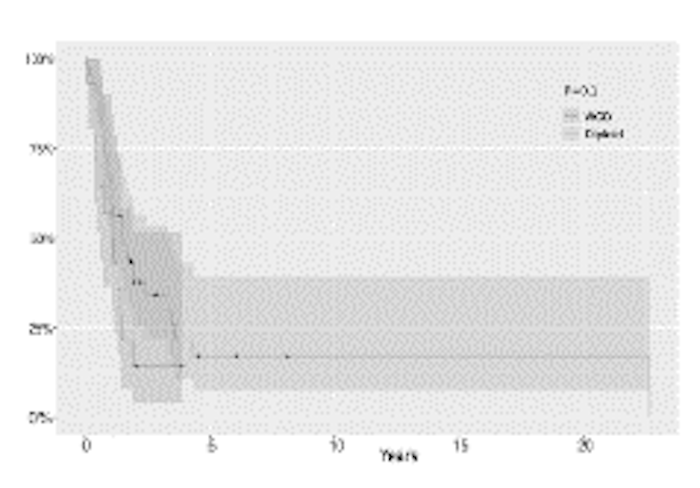
from 60 DIPG patients. This revealed 25% DIPGs acquired whole-genome duplications (WGD) early during
tumor evolution. WGD samples are associated with loss of TP53 and poorer survival. In addition, almost all
WGD samplers harbor complex structural variations (SVs) and show characteristic short microhomology at SV
breakpoints. Mutation analysis revealed that H3K27M driver mutation is acquired early during tumor clonal
evolution. Mutation signature analysis identified a unique mutational process at a late stage of tumor evolution.
This study revealed that tumor evolution of DIPG is characterized by chromosomal instability shaped by DNA
repair defects and dynamic mutational processes. Our work shed new insights on the disease pathogenesis of
DIPG and provided rationale for designing novel therapy for this deadly disease.
Acknowledgements
We thank members of Children’s Brain Tumor Tissue Consortium (CBTTC) (www.cbttc.org) for their support of open access, biospecimen driven research.

