Epigenetic Drivers in Medulloblastoma

Alexandros Tzatsos
CBTN Data
Backer
1R01CA222930-01A1
About this
Project
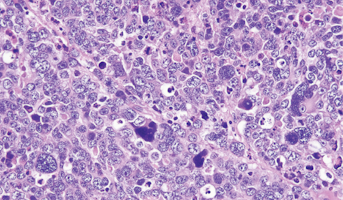
Medulloblastoma is a malignancy of the cerebellum, and is the most common pediatric brain tumor. Current molecular classifications stratify tumors into four subgroups: wingless (WNT), sonic hedgehog (SHH), Group 3, and Group 4. Patients within these groups align in their age demographics, prognosis, risk of metastatic disease, and response to standard therapy. The WNT and SHH subgroups harbor mutations in their namesake pathways, and have a relatively good prognosis as they rarely develop metastases. However, the ambiguously named Group 3 and 4 tumors are poorly characterized on the molecular level and have worse prognosis as they frequently develop metastatic disease. It is imperative to have a better molecular understanding of the underlying causes and drivers of these tumors. Researchers have previously generated epigenetic maps of the aforementioned subgroups and found profound alterations on the enhancer chromatin landscape. Chromatin refers to the bundles of proteins, RNA, and DNA that make up chromosomes. Researchers will analyze genetic data provided through the Pediatric Brain Tumor Atlas for alterations that may affect organization of such enhancer chromatin in the different Medulloblastoma subgroups.
Ask The
Scientists
What are the goals of this project?
We generated epigenetic maps of the aforementioned subgroups and found profound alterations on the enhancer chromatin landscape. Here, we seek to employ WGS to identify copy number and structural alterations in the non-coding genome that may drive remodeling of enhancer chromatin.
We will analyze WGS data for copy number and other structural alterations that may affect organization of enhancer chromatin in the different Medulloblastoma subgroups.
What is the impact of this project?
Researchers aim to develop a better molecular understanding of the most aggressive medulloblastoma subtypes in an effort to develop targeted therapeutics.
Why is the CBTN request important to this project?
With a better understanding of medulloblastoma subgroup drivers, researchers can work to improve diagnostics, prognostics, and therapies for this disease.
Project
Results
The Pediatric Brain Tumor Atlas provides researchers with high quality data on medulloblastoma subtypes needed to complete this work.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing access to whole genome sequencing data for medulloblastomas.