Impeding LIN28 Function in ATRT
Email Principal Investigator

Giselle Saulnier Sholler
CBTN Specimen
CBTN Participants
CBTN Pre-clinical Models
Backer
Donor funds
About this
Project
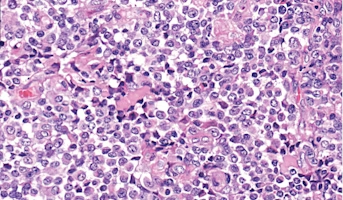
Atypical teratoid rhabdoid tumors (ATRT) are a pediatric central nervous system (CNS) tumor that accounts for 30% of CNS tumors in patients younger than 3 years old. Genomic profiling has suggested molecular differences within ATRT, and three genomic subtypes have been described. (Group1-TYR, Group2A-SHH and Group2B-MYC) However, studies investigating ATRT have used a limited number of cell lines that have not represented all subtypes of this tumor. Better understanding of this complex disease and developing novel treatment approaches are desperately needed. The first aim of this specific project is to determine if pharmacological treatment of ATRT cell lines with difluoromethylornithine (DFMO) results in positive changes. The second aim of this project is to figure out if DFMO induced effects occur in all subtypes of ATRT, or if responses are subtype dependent. Information learned from these experiments may lead to the identification of biomarkers predictive of response to DFMO treatment and the development of clinical trials testing the efficacy of DFMO in ATRT patients.
Utilizing the resources provided by the Children’s Brain Tumor Network will greatly improve the impact of the experiments being undertaken in this project.
Ask The
Scientists
What are the goals of this project?
Researchers will determine whether the use of DFMO has positive results in the treatment of ATRT, and whether drug response varies across tumor subtype.
What is the impact of this project?
This project could set the groundwork for clinical trials into the use of DFMO in ATRT patients.
Why is the CBTN request important to this project?
Previous studies have been held back by the lack of samples comprising all known ATRT subtypes. The Children’s Brain Tumor Network provides researchers with the variety of samples needed to complete this work.
Specimen Data
The Children's Brain Tumor Network will contribute to this project by providing ATRT cell lines.