
Javad Nazarian
CBTN Data
Backer
Hyundai Hope on Wheels Foundation Grant
Swim Across America Young Investigator Award
Gary Fegel Foundation Donation
About this
Project
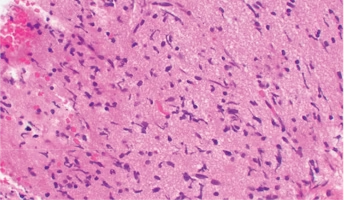
Diffuse intrinsic pontine glioma (DIPG) remains a fatal disease and the identification and development of new treatment approaches are greatly needed. A novel computational-experimental Cancer Systems Biology approach involves identifying the interactions of master regulator (MR) proteins, with mutations and their influence on cancer development. This approach may also predict and prioritize drugs that reverse the effects of these interactions. This study will apply such an approach to evaluate new treatment strategies for patients with DIPG. Researchers will analyze the Pediatric Brain Tumor Atlas and other DIPG gene expression data using innovative computational methods. This analysis will describe the interactions of MR proteins in DIPG tumors and prioritize drugs by their ability to shut down the most aberrant MR proteins. This method can be targeted to individual patient samples and across the disease as a whole allowing for subtyping of tumors to occur while characterizing DIPG gene regulatory networks overall. This project will evaluate the efficacy of treatments predicted by the MR approach in patient-derived DIPG cell lines and animal models. This will advance drug development and suggest novel therapy approaches in DIPG to improve outcomes in this difficult-to-treat disease.
Ask The
Scientists
What are the goals of this project?
The main goal of this project is to advance drug and therapy development for DIPG patients through the analysis of master regulator proteins, identify subtypes of DIPG for improved therapeutics.
What is the impact of this project?
This research uses an approach that will allow for the prioritization of drugs and therapies for patients with DIPG in an effort to improve survival outcomes, provide the community a understanding of the interactions occurring between MR proteins and mutations in DIPG and provide a better understanding of subtypes of DIPG.
Why is the CBTN request important to this project?
The Pediatric Brain Tumor Atlas provides researchers with rare gene expression data from DIPG patients.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing access to the Pediatric Brain Tumor Atlas.
Meet The
Team
Jovana Pavisic
Stergios Zacharoulis
Andrea Califano
Peter Canoll
Paul Kirk Reardon