Development of Preclinical Models of Pediatric Ependymoma
Email Principal Investigator
About this
Project
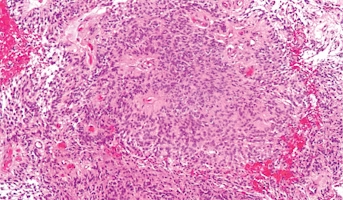
Strong preclinical models, used to mimic conditions of ependymoma growth in the human body, are essential to the development of targeted therapies. Such models allow researchers to study the tumors as well as perform drug screening before testing therapies on a patient. Ependymoma are known to be difficult to grow, but researchers have found recent success utilizing a novel technique to establish long term 3D cultures of this nervous system tissue. Frozen supratentorial ependymoma (ST-EPN) specimens were provided by CBTN for this project and will be used to create up to 10 models, advancing the development of new treatments.
Ask The
Scientists
What are the goals of this project?
An array of preclinical models of ependymoma will be produced for use by researchers in drug screening.
What is the impact of this project?
Strong preclinical models for ependymoma will pave the way for researchers to develop and test new therapeutic options.
Why is the CBTN request important to this project?
CBTN has the largest available collection of pediatric brain cancer samples and will provide frozen specimens integral for the advancement of this research.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing tissue in freezing media for the generation of these models.
Meet The