About this
Project
Children with high-grade glioma, diffuse intrinsic pontine glioma, rhabdoid brain tumors, and certain other types of brain tumors experience a low likelihood of survival even with the maximum available therapy. The standard therapies for pediatric brain tumors, including surgery, intensive chemotherapy, and radiation therapy are often toxic for patients, therefore there is a strong need to identify, develop, and test new therapies. Preclinical models are a common way for researchers to study tumor types as well as test diagnostics and therapies. An avenue for the development of new therapies is the identification of biomarkers, indicators or targets in the body that can help predict patient response to certain treatments. Researchers will use data available through the Pediatric Brain Tumor Atlas to search for such biomarkers. This information can then be used in the development of combination therapies tested on animal models, allowing more accurate predictions of a patient’s reaction to treatment, reducing toxicities.
Ask The
Scientists
What are the goals of this project?
The goals of this project are to identify biomarker targets in high grade pediatric brain cancers that will then be used for the development of combination therapies tested on models.
What is the impact of this project?
Once relevant biomarker targets are identified, researchers can develop and test combination therapies on models, giving more accurate predictions of how patients may respond to treatments.
Why is the CBTN request important to this project?
Interrogation of extensive data on pediatric brain cancers is necessary to find usable biomarkers and the Pediatric Brain Tumor Atlas provides researchers with the information they need to push the development of new treatments forwards.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing access to the Pediatric Brain Tumor Atlas.
related
Histologies

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
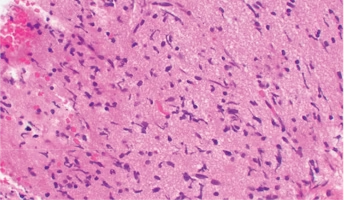
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with

