Therapeutic Targeting of the Blood-Brain Barrier in Pediatric Malignant Brain Tumors
Email Principal Investigator

Sadhana Jackson
CBTN Data
CBTN Specimen
CBTN Participants
CBTN Pre-clinical Models
About this
Project
Glioblastoma is an extremely aggressive brain tumor that has poor outcomes despite the use of multimodality therapy. Treatment options for malignant gliomas are significantly limited by the presence of the blood-brain barrier (BBB). The BBB is a protective element of the central nervous system that keeps toxins or pathogens circulating in the blood away from the brain. The protective nature of the BBB in turn also inhibits effective therapy from reaching tumor cells. The majority of agents used to combat high grade glioma growth have agents known to be able to cross the BBB. However, there are many agents that are effective against malignant glioma, but are not being utilized because they fail to cross the BBB. Due to this, there is a strong interest to enhance delivery of certain agents in hopes of improving survival rates of patients with malignant brain tumors. Additionally, the diversity in BBB integrity aids glioma stem cells (GSCs) in evading treatment, proliferating and developing treatment resistance, despite multi-modality therapy. The failure of current therapies to eliminate specific GSC subpopulations has been considered a major contributing factor to tumor recurrence. The ultimate goal of this research is to enhance therapies targeting malignant tumor cells and their supportive BBB, in an effort to improve treatment response and prolong overall survival. The Children’s Brain Tumor Network provides researchers on this project access to a large database of samples they are not able to access elsewhere.
Ask The
Scientists
What are the goals of this project?
Researchers seek to enhance therapeutic options for patients with glioblastoma by better understanding the BBB tumor environment and drug interactions with the BBB.
What is the impact of this project?
Enhancing the delivery of therapeutics through the BBB will lead to improved treatment response and overall survival of patients with glioblastoma.
Why is the CBTN request important to this project?
This work requires rare specimens and data made accessible through the Pediatric Brain Tumor Atlas.
Specimen Data
The Children's Brain Tumor Network will contribute to this project by providing cell lines and patient derived xenografts.
related
Histologies

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
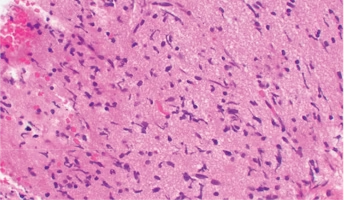
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with


