Sriram Venneti

About
Associate Professor
UNIVERSITY OF MICHIGAN
Dr. Venneti’s lab studies the biology of pediatric and adult brain tumors (such as gliomas thought to be of glial origin) with a specific focus on understanding the regulation of cancer metabolism and epigenetics as drivers of pathology. Cancer cells including brain tumor cells reprogram their metabolism by altering nutrient uptake and metabolism to support their aberrant proliferation and survival. The two principle nutrients that cancer cells use are the sugar glucose and the amino acid glutamine. These two nutrients are central to many anabolic processes including the biosynthesis of ATP, nucleotides, proteins and lipids. Recent years have seen the emergence of the concept that metabolic reprogramming in cancer cells is not a passive process. Rather, oncogenes and inactivated tumor suppressors in cancer cells directly reprogram the metabolism of cancer cells. These metabolic pathways can directly rewire the epigenome and provide novel avenues to develop newer diagnostic and therapeutic targets.

Michigan Medicine C.S. Mott Children’s Hospital
research
Interests

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
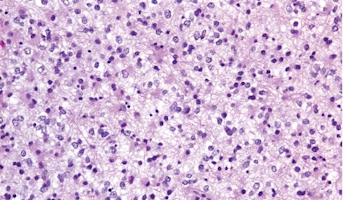
Low-Grade Glioma
Low-Grade Gliomas also called astrocytomas are the most common cancer of the central nervous system in children. They represent a heterogeneous group of tumors that can be discovered anywhere within the brain or spinal cord. Although surgical resection may be curative, up to 20% of children still su