Development of the Ganglioside GD2 as a Biomarker and Clinical Trial Endpoint for Childhood Cancers
Email Principal Investigator

Frank M. Balis
CBTN Specimen
CBTN Participants
CBTN Samples
Backer
COGSM-ITSC Grant
Completed
About this
Project
GD2 is a molecule expressed on the surface of tumors such as high grade glioma or medulloblastoma. and GD2 has many of the characteristics of an ideal biomarker. Biomarkers can be used as therapeutic targets for the treatment of cancer and can also serve to provide accurate prognostics and predict treatment response. The goal of this project is to determine GD2 concentrations in patients with a variety of common childhood cancers, including brain tumors, and in children without cancer. Researchers hypothesize that GD2 will not be quantifiable in the serum of patients with most common types of childhood cancer and children without cancer and that GD2 is a tumor-specific biomarker for neuroblastoma and brain tumors. The Children's Brain Tumor Network contributed to this project by providing plasma samples that will be screened for the presence of GD2. Validation of GD2 as a tumor biomarker opens the door to new therapies and could help to expedite clinical trial processes leading to faster development.
Ask The
Scientists
What are the goals of this project?
Researchers seek to validate GD2 as a tumor biomarker through the analysis of plasma samples.
What is the impact of this project?
If validated, GD2 based therapies verified by this project could lead to the acceleration of clinical trial processes, thus bringing novel therapies to patients as quickly as possible.
Why is the CBTN request important to this project?
The Children’s Brain Tumor Network will support this project through the provision of serum samples from across pediatric brain tumor types.
Project
Results
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing plasma samples.
related
Histologies
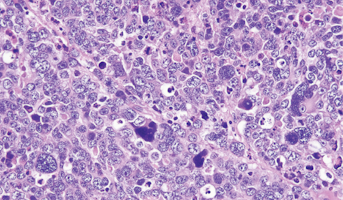
Medulloblastoma
Medulloblastomas comprises the vast majority of pediatric embryonal tumors and by definition arise in the posterior fossa, where they constitute approximately 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% or less of all childhood brain tumors.The clinical feature

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test


