Investigating Disease Mechanism In Adamantinomatous Craniopharyngioma Using New Cell Line Models
Email Principal Investigator
About this
Project
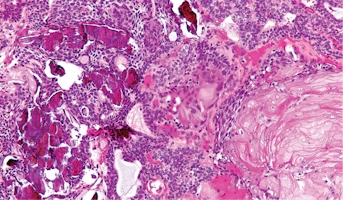
Adamantiomatious craniopharyngioma (ACP) is an intracranial tumor that is histologically benign, but the clinical manifestation of the disease can have profound effects due to the location of the tumor. Current understanding of the disease pathology is limited and there are no targeted drug therapeutics for the disease. The lack of reliable cell line models for ACP has limited researcher’s ability to utilize techniques to study disease mechanisms and drug interactions. In this study, researchers propose to characterize and validate the new ACP tumor cell lines developed and provided by the Children’s Brain Tumor Network. The goal of this project is to characterize and validate the cell line model for use as reliable in-vitro models of ACP. Once established, this model can be utilized to great extent in the laboratory to explore disease mechanisms and drug interactions of ACP. Access to matched samples and curated database of clinical, histological, and molecular information associated with these cell lines that is available from the Children’s Brain Tumor Network is very critical for the success of this project.
Ask The
Scientists
What are the goals of this project?
This project will serve as validation for the use of ACP models, to be used to better understand this tumor.
What is the impact of this project?
With quality models for ACP established, researchers will then be able to explore disease mechanisms and drug interactions in the pursuit of targeted therapies.
Why is the CBTN request important to this project?
Not only does the Children’s Brain Tumor Network provide researchers with high quality specimens, but the specimens are matched with important data that makes this work more efficient.
Specimen Data
The Children's Brain Tumor Network is contributing to this project by providing cell lines.
Meet The
Team
Institutions

Children's Hospital of Colorado

Primary
Operations Center

Children’s Hospital of Philadelphia
Joined onOperations Center for the Children’s Brain Tumor Tissue Consortium, the Children’s Hospital of Philadelphia (CHOP) is currently ranked 1st nationally for their Pediatric Cancer Program by U.S. News & World Report. CHOP’s Biobank is home to the CBTTC’s pediatric brain and CNS tumor biorepository; the