Cassie Kline

About
Director of Clinical Research in the Department of Neuro-Oncology at the Children's Hospital of Philadelphia
Children's Hospital of Philadelphia
Pediatric Neuro-Oncology, Clinical research, Early phase clinical trials in neuro-oncology, Developmental therapeutics

Children’s Hospital of Philadelphia
scientific
Projects

Specimen
Planning
Investigating Disease Mechanism In Adamantinomatous Craniopharyngioma Using New Cell Line Models
Adamantiomatious craniopharyngioma (ACP) is an intracranial tumor with no currently available targeted drug therapeutics. Using cell lines provided by the Children’s Brain Tumor Network, researchers will develop new models for use in identifying therapeutic opportunities for this disease.
Craniopharyngioma

Todd Hankinson

Data
Planning
Precision Imaging of Pediatric High-grade Gliomas with Quantitative Diffusion Weighted Imaging and Texture Analysis to Identify Imaging Biomarkers That Predict Tumor Genetics and Patient Outcomes
The recently characterized molecular marker, histone H3 K27M, could prove important to the treatment of pediatric gliomas. Researchers will interrogate data from the Pediatric Brain Tumor Atlas in order to better understand this new class of malignant gliomas.
HGG, DIPG

Mariam Aboian
Support
Planning
Diversity, Equity, and Inclusion: Translation of Informed Consent Forms
In November 2020, the FDA released guidance to help promote clinical trial enrollment practices that could lead to clinical trials that better reflect diverse populations who may benefit from treatments, medical devices, or drugs that are being investigated. Despite previous efforts, there remain gaps in inclusion of under-represented populations in many clinical trials. One of the barriers to being inclusive among all groups are the languages that are offered for informed consent practices. Currently, the Children’s Hospital of Philadelphia and collaborating institutions typically offer multilingual staff and informed consents in limited languages. But, the options available at each site vary and can be costly and burdensome at the level of each individual institution/clinical research team. Our goal is to remove this potential barrier by providing consortium level informed consents and study documents in multiple languages to ensure enrollment and recruitment of under-represented minorities (URM) in languages understood by a diverse population of patients and families. By providing these necessary resources, non-English speaking patients and families will be afforded the same opportunity to understand and participate in clinical trial enrollments as English-speaking patients and families.
All Brain Tumor Types

Cassie Kline
research
Interests
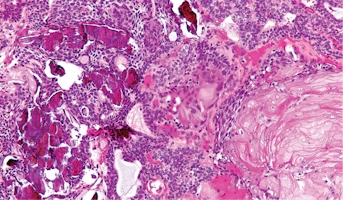
Craniopharyngioma
Childhood craniopharyngiomas are rare tumors usually found near the pituitary gland (a pea-sized organ at the bottom of the brain that controls other glands) and the hypothalamus (a small cone-shaped organ connected to the pituitary gland by nerves).Craniopharyngiomas are usually part solid mass and

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
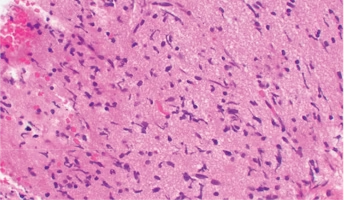
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with