Precision Imaging of Pediatric High-grade Gliomas with Quantitative Diffusion Weighted Imaging and Texture Analysis to Identify Imaging Biomarkers That Predict Tumor Genetics and Patient Outcomes
Email Principal Investigator
About this
Project
The current standard of care for treatment of pediatric patients with high grade gliomas is surgery followed by radiotherapy with concurrent chemotherapy. Midline gliomas with histone H3 K27M mutation are a new class of malignant gliomas that present specific challenges. These challenges include having extremely poor prognosis due to being located in regions of the brain where surgery is difficult. Pediatric high grade gliomas are molecularly different from adult gliomas and in comparison to adults, children with high grade gliomas have persistently poor clinical outcomes. Molecular markers have been shown to be important for treatment of adult gliomas. The recently characterized molecular marker, histone H3 K27M, could prove similarly important in the treatment of pediatric gliomas. Due to invasive potentially site-limited availability of molecular testing, there is a critical need to identify imaging biomarkers of pediatric high grade gliomas. Researchers are looking for biomarkers that could predict tumor molecular markers used as therapy targets, tumor response to radiotherapy and chemotherapy, and clinical performance levels of children undergoing treatment. The goal of the proposed study is to identify imaging markers that predict the molecular markers, histologic features, and clinical outcomes in a cohort of pediatric patients with high grade gliomas. Researchers will do this using data from patients that underwent tissue sampling, genetic analysis of tumor tissue, and at least a 12 month follow up, available through the Pediatric Brain Tumor Atlas.
Ask The
Scientists
What are the goals of this project?
The goal of this project is to deepen the understanding of the malignant form of glioma, midline gliomas with histone H3 K27M mutations.
What is the impact of this project?
Researchers are hoping to find imaging biomarkers that could be used as therapy targets, predict tumor response to radiotherapy and chemotherapy, and increase clinical performance levels of children undergoing treatment.
Why is the CBTN request important to this project?
Researchers require data from patients that underwent tissue samples, genetic analysis of that tissue, and had a 12 month follow up, data made available only through the Pediatric Brain Tumor Atlas.
Meet The
Team
Dr. Soonmee Cha , UCSF

New Haven, CT, USA

Philadelphia, PA, USA

San Francisco and Oakland, CA, USA
Institutions

Yale School of Medicine

Primary
Operations Center

Children’s Hospital of Philadelphia
Joined onOperations Center for the Children’s Brain Tumor Tissue Consortium, the Children’s Hospital of Philadelphia (CHOP) is currently ranked 1st nationally for their Pediatric Cancer Program by U.S. News & World Report. CHOP’s Biobank is home to the CBTTC’s pediatric brain and CNS tumor biorepository; the

Primary

UCSF Benioff Children's Hospital
Joined onThe Pediatric Brain Tumor Center at UCSF Benioff Children’s Hospital, with campuses in San Francisco and Oakland, harnesses the power of world-class experts from all disciplines related to child brain health, including neurology, neurosurgery, neurocritical care, neurogenetics, neuro-oncology, rehab
related
Histologies

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
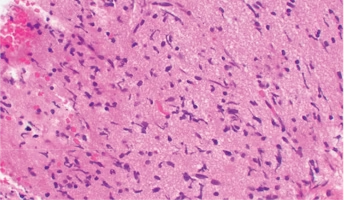
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with
