Eric Raabe

About
Associate Professor of Oncology; Instructor of Pathology
The Johns Hopkins Hospital
A single pivotal experience that occurred years ago influenced Dr. Eric Raabe’s decision to become a pediatric oncologist.
Dr. Raabe was an undergraduate student volunteering at a children’s hospital. He had gotten to know an adolescent pediatric cancer patient during his volunteer stint, and vividly recalls a time when the boy appeared particularly sad. The shades in his room were shut tight. He sat in his bed playing video games and wasn’t much interested in talking. He was awaiting the results of a biopsy, which would tell him whether or not he had relapsed while receiving therapy. A relapse likely would signify a very poor outcome for the young man. The results came back negative. The boy’s mood immediately brightened, and he asked for the shades to be open and a pizza ordered, with everything on it.
“In that moment, I thought to myself: Here is what pediatric cancer doctors do--help a sick, scared patient to get better,” said Dr. Raabe. “I love supporting patients and families through the dark times and celebrating the fun times with them.”
Dr. Raabe knows well the need for supporting patients and their families, as he specializes in some of the most high-risk pediatric brain cancers: MYC-driven medulloblastoma (group 3), diffuse intrinsic pontine glioma (DIPG), atypical teratoid rhabdoid tumor (AT/RT), and glioblastoma (GBM). Currently, there are very few long-term survivors of these diseases. The need for new treatments aimed at these cancers is critical.
Dr. Raabe explains that, despite becoming more clever in how drugs and radiation are used, pediatric oncologists have discovered very few new drugs in the past 30 years. “We are using 1980s therapy to treat patients today,” Dr. Raabe says. He and his associates are working hard to change that.
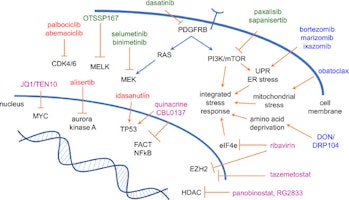
Dr. Raabe describes a new approach that he and his colleagues are taking in an attempt to attack aggressive pediatric brain tumors. “We are targeting abnormal metabolism in pediatric brain tumors. Some of the genetic alterations that drive the growth of these tumors make the metabolism of tumor cells much different from normal cells. We can use metabolic inhibitors to kill these cells in the laboratory and in mouse experiments. The next step for both medulloblastoma and AT/RT will be clinical trials,” Dr. Raabe explains.
While the current lack of effective and long-term treatments for these aggressive cancers is frustrating, the wave of potential new therapies offers promise.

Johns Hopkins Medicine
research
Interests
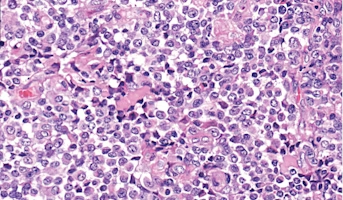
Atypical Teratoid/Rhabdoid Tumor
Central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT) is a very rare, fast-growing tumor of the brain and spinal cord. It usually occurs in children aged three years and younger, although it can occur in older children and adults. About half of these tumors form in the cerebellum or
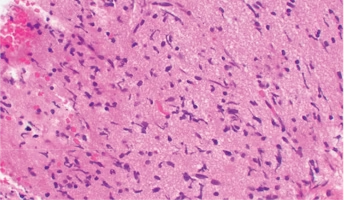
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with