GPC2 as an Immunotherapeutic Target in Medulloblastoma and other Pediatric Brain Tumors
Email Principal Investigator

Kristopher Bosse
CBTN Specimen
CBTN Participants
CBTN Samples
CBTN Pre-clinical Models
Backer
Alex's Lemonade Stand Foundation
Damon Runyon
HHOW
Iron Matt Foundation
About this
Project
Medulloblastoma and other pediatric brain cancers could be candidates for immunotherapy, a therapy that utilizes a patient’s own immune system to attack a tumor. Immunotherapy requires molecular targets and medulloblastomas and other brain tumors differentially express cell surface molecules that could act as targets. The recently FDA-approved chimeric antigen receptor (CAR) T cell therapy (a type of immunotherapy) for acute lymphoblastic leukemia, pioneered at the Children's Hospital of Philadelphia, provides proof that immunotherapies can combat pediatric cancers. Challenges remain in identifying molecules that meet optimal immunotherapeutic safety and efficacy criteria for use in the pediatric patient population. Researchers have recently discovered that the signaling co-receptor glypican-2 (GPC2) meets these criteria in both medulloblastomas and neuroblastomas. GPC2 is a highly expressed medulloblastoma and neuroblastoma cell surface molecule that is not detectable at significant levels on normal pediatric tissues. The group has developed a GPC2-directed antibody-drug conjugate (ADC) that potently inhibits the growth of neuroblastoma in animal models. However, the efficacy of this ADC in medulloblastoma models remains undefined. Researchers have also found that GPC2 may be overexpressed in other lethal pediatric brain tumors, such as high-grade gliomas (HGGs), peripheral neuroectodermal tumors (PNETs) (including the universally lethal embryonal tumors with multilayered rosettes [ETMRs]), and atypical teratoid rhabdoid tumors (ATRTs). The central hypothesis of this project is that targeting GPC2 with the identified ADC in medulloblastoma and other GPC2-expressing pediatric brain tumors will lead to better outcomes for patients. The Children's Brain Tumor Network contributed cell lines and tissue in freezing media necessary to develop the models needed for this line of research.
Ask The
Scientists
What are the goals of this project?
The main goal of this project is to determine whether targeting GPC2 is a viable option for the immunotherapeutic treatment of various pediatric brain cancers.
What is the impact of this project?
Researchers hypothesize that if proven a successful therapeutic target, GPC2 related therapies could lead to much better outcomes for pediatric brain cancer patients.
Why is the CBTN request important to this project?
The Children’s Brain Tumor Network provided cell lines and high quality frozen specimens integral to this research.
Specimen Data
The Children's Brain Tumor Network contributed cell lines and tissue in freezing media for in vitro studies.
Meet The
Team
related
Histologies
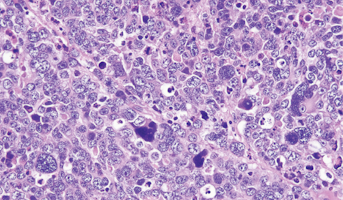
Medulloblastoma
Medulloblastomas comprises the vast majority of pediatric embryonal tumors and by definition arise in the posterior fossa, where they constitute approximately 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% or less of all childhood brain tumors.The clinical feature

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
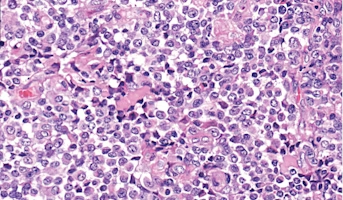
Atypical Teratoid/Rhabdoid Tumor
Central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT) is a very rare, fast-growing tumor of the brain and spinal cord. It usually occurs in children aged three years and younger, although it can occur in older children and adults. About half of these tumors form in the cerebellum or



