MET Alterations in DMGs
Email Principal Investigator

Payal Jain
CBTN Specimen
CBTN Participants
CBTN Pre-clinical Models
Backer
St. Baldrick's Foundation
About this
Project
Diffuse midline gliomas (DMGs) are pediatric brain tumors accounting for 10-15% of all childhood brain tumors. Researchers have analyzed DMG data including data from the Pediatric Brain Tumor Atlas and found the mesenchymal-epithelial transition factor (MET) to be dysregulated in many cases of DMGs. The MET is a process by which stem cells become epithelial cells, surface cells such as cells found in the skin, organs, and vessels. The mechanisms driving MET dysregulation in these tumors remains largely unknown, but recent research is pointing towards gene fusions as a driver. Gene fusions (GFs) occur when two previously independent genes fuse. GFs have been increasingly recognized as successful targets in the treatment of many cancers, but their prevalence and therapeutic use remains understudied in DMGs. Recent findings by this research group provide a new context for studying MET biology and exploring MET gene fusions as personalized therapeutic targets in DMGs. Over the past several years, the researchers on this project have highlighted the recurrence of GFs that drive oncogenic processes in pediatric low-grade gliomas (pLGGs) and reported their potential to guide diagnosis, prognosis and/or therapy. Through better understanding of how MET biology contributes to DMGs, researchers could unlock new ways to use MET as a therapy target for the treatment of such tumors, expanding the scope of precision medicine targets in DMGs.
Ask The
Scientists
What are the goals of this project?
The main research goal of this project is to better understand how MET biology contributes to DMGs development.
What is the impact of this project?
A deeper understanding of the impact of MET dysregulation on DMGs could lead to new ways to use MET as a therapy target for DMGs and related tumors.
Why is the CBTN request important to this project?
The Children’s Brain Tumor Network’s support of this project includes the provision of rare, high quality cell lines for use by researchers.
Specimen Data
The Children's Brain Tumor Network is contributing to this project by providing cell lines.
Meet The
Team
Institutions

Primary
Operations Center

Children’s Hospital of Philadelphia
Joined onOperations Center for the Children’s Brain Tumor Tissue Consortium, the Children’s Hospital of Philadelphia (CHOP) is currently ranked 1st nationally for their Pediatric Cancer Program by U.S. News & World Report. CHOP’s Biobank is home to the CBTTC’s pediatric brain and CNS tumor biorepository; the

Satellite
Michigan Medicine C.S. Mott Children’s Hospital
C.S. Mott Children’s Hospital’s pediatric cancer specialists offer special expertise in caring for children with relapsed and resistant cancers. Michigan Medicine is home to the largest pipeline of Phase 1 clinical trials in the state, with both Michigan-led and multi-site clinical trial offerings.I

National Institutes of Health
related
Histologies

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
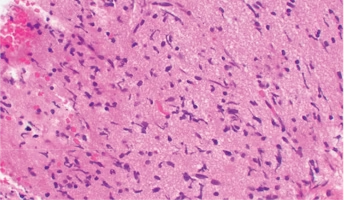
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with


