
Accelerating childhood brain tumor research to faster cures
Over 30 types of pediatric brain and spinal cord tumor clinical and molecular data, biospecimens, and cell-lines are available at no cost to academic researchers. Our open science model has shortened research time by up
to 20 years.
By working together to collect and share resources, we can save more children.

CBTN by the Numbers
Brain and CNS tumors are the most common cause of disease related death in children aged 0–19 years in the U.S. and across the globe, with approximately 412,000 children and young adults living with a brain tumor each year.
featured
Scientific Projects

Specimen
Ongoing
Gene Expression Analysis Platform Evaluation for FFPE Specimen Material-Based Studies
Newly developed technology is advancing the ability of researchers to gain important insight from small clinical specimens.
Medulloblastoma, HGG

Mateusz Koptyra
Data
Specimen
Ongoing
Proteogenomic Identification of Structural Variations
Changes to DNA that can give rise to cancers often create fusion proteins, proteins that may be useful as therapeutic targets. Using data from the Pediatric Brain Tumor Atlas, researchers will analyze for the presence of such proteins in an effort to advance treatment options for pediatric brain cancers.
Medulloblastoma, DIPG

Brian Rood

Data
Ongoing
Deciphering the Molecular Characteristics of Pediatric Meningiomas
Pediatric meningiomas are extremely rare tumors and are often of an aggressive form unresponsive to available treatments. Specimens from the Pediatric Brain Tumor Atlas will allow researchers to study this rare tumor in an effort to develop targeted therapies.
Medulloblastoma, HGG, LGG, Ependymoma, Meningioma, Schwannoma

Nadia Dahmane
cbtn
Data Science
featured
Available Resources
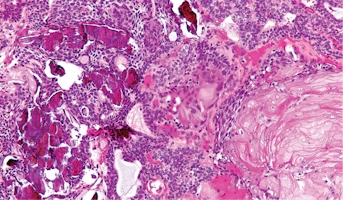
Craniopharyngioma
Childhood craniopharyngiomas are rare tumors usually found near the pituitary gland (a pea-sized organ at the bottom of the brain that controls other glands) and the hypothalamus (a small cone-shaped organ connected to the pituitary gland by nerves).Craniopharyngiomas are usually part solid mass and
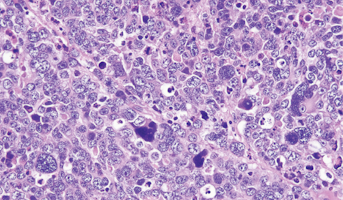
Medulloblastoma
Medulloblastomas comprises the vast majority of pediatric embryonal tumors and by definition arise in the posterior fossa, where they constitute approximately 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% or less of all childhood brain tumors.The clinical feature

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test


