Deciphering the Molecular Characteristics of Pediatric Meningiomas
Email Principal Investigator
About this
Project
Meningiomas are the most common tumors occurring in the brain and spine. About 80% of them show benign features responsive to surgery alone. 20%, however, require extensive treatment including repeat surgery, radiation, and/or chemotherapy, and in many cases the tumors still recur. Pediatric meningiomas, however, are extremely rare tumors and are often of the more aggressive form. It is thought that the underlying biology of pediatric meningiomas is different from their adult counterparts, and this study seeks to investigate those differences. Whole Genome Sequencing (WGS), which identifies the base chemicals that make up DNA, will be carried out on specimens from the Pediatric Brain Tumor Atlas. Researchers aim to gain a deeper understanding of pediatric meningiomas that could establish new therapies designed specifically for the pediatric population.
Ask The
Scientists
What are the goals of this project?
Pediatric meningiomas are not as well understood at adult meningiomas and researchers in this project seek to close that knowledge gap in the pursuit of targeted therapies.
What is the impact of this project?
Through addressing the differences between adult and pediatric meningiomas, researchers are paving the way for the development of new targeted therapies for pediatric populations. The data generated will help characterize this tumor type and be shared with the research community.
Why is the CBTN request important to this project?
Pediatric meningiomas are rare tumors, but the Pediatric Brain Tumor Atlas will give researchers access to the specimens needed to carry out this research.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing access to the Pediatric Brain Tumor Atlas
Meet The
Team

New York, NY, USA

Philadelphia, PA, USA

Philadelphia, PA, USA
Institutions

Primary

Weill Cornell Medicine
Joined onThe Weill Cornell Medicine Pediatric Brain and Spine Center, located on the Upper East Side campus of New York-Presbyterian/Weill Cornell Medical Center, is nationally recognized for its leadership in the treatment of disorders of the central nervous system in children, particularly brain and spinal

Primary
Operations Center

Children’s Hospital of Philadelphia
Joined onOperations Center for the Children’s Brain Tumor Tissue Consortium, the Children’s Hospital of Philadelphia (CHOP) is currently ranked 1st nationally for their Pediatric Cancer Program by U.S. News & World Report. CHOP’s Biobank is home to the CBTTC’s pediatric brain and CNS tumor biorepository; the
related
Histologies
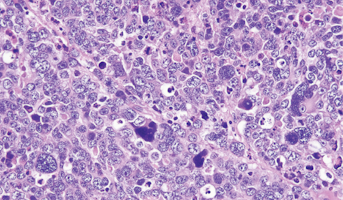
Medulloblastoma
Medulloblastomas comprises the vast majority of pediatric embryonal tumors and by definition arise in the posterior fossa, where they constitute approximately 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% or less of all childhood brain tumors.The clinical feature

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
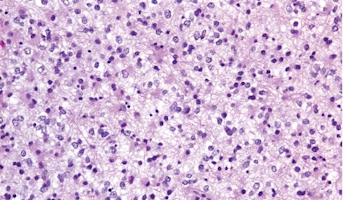
Low-Grade Glioma
Low-Grade Gliomas also called astrocytomas are the most common cancer of the central nervous system in children. They represent a heterogeneous group of tumors that can be discovered anywhere within the brain or spinal cord. Although surgical resection may be curative, up to 20% of children still su
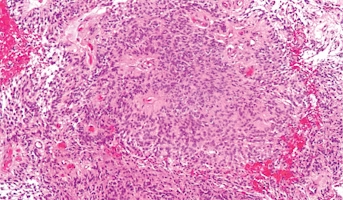
Ependymoma
Ependymomas arise from ependymal cells that line the ventricles and passageways in the brain and the center of the spinal cord. Ependymal cells produce cerebrospinal fluid (CSF). These tumors are classified as supratentorial or infratentorial. In children, most ependymomas are infratentorial tumors
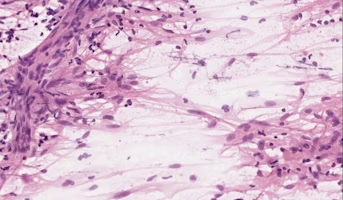
Meningioma
A meningioma is a primary central nervous system (CNS) tumor. This means it begins in the brain or spinal cord. Overall, meningiomas are the most common type of primary brain tumor. However, higher grade meningiomas are very rare.
Schwannoma
Schwannoma is a rare type of tumor that forms in the nervous system. Schwannoma grows from cells called Schwann cells. Schwann cells protect and support the nerve cells of the nervous system. Schwannoma tumors are often benign, which means they are not cancer. But, in rare cases, they can become can


