Mariella Filbin

About
Research Co-Director, Pediatric Neuro-Oncology Program; Assistant Professor of Pediatrics, Harvard Medical School; Physician
Dana-Farber Cancer Institute
Dr. Mariella Filbin is the Research Co-Director at Harvard Medical School. She received her MD and PhD in biochemistry and molecular biology from the Medical University of Graz, Austria. Following graduation she completed her pediatric residency at the Boston Children's Hospital and completed a fellowship in pediatric hematology/oncology at Dana-Farber/Boston Children's Cancer and Blood Disorders Center. She joined the faculty at Dana Farber/Boston Children's as
a pediatric neuro-oncologist in July 2017.
Dr. Filbin's research is focused establishing cellular networks of tumor dependencies in pediatric brain tumors with the goal of finding new targeted therapies.

Harvard Medical School
scientific
Projects

Data
Ongoing
Analysis of Chromatin Pathways as Regulators of High Grade Glioma Gene Expression Patterns
Comprehensive research into high grade gliomas is necessary to find new effective therapies. Using the Pediatric Brain Tumor Atlas, researchers will identify patterns in gene expression that could pave the way for the development of new therapeutics.
HGG, DIPG

Jamie Anastas

Specimen
Ongoing
Discovering Novel Epigenetic Dependencies in Pediatric High-grade Glioma
Primary resistance to chemotherapy is a major challenge in treating brain tumors today. Finding novel and specific tumor dependencies is therefore essential for effective drug development and improving patient outcomes.High-grade gliomas (HGG) are the most aggressive brain tumors in children and you
HGG, DIPG

Mariella Filbin

Data
Ongoing
PDGFRA expression in K27M gliomas
A subset of high grade glioma (HGG) tumors are known to have a specific mutation called the H3K27M mutation. Researchers on this project will analyze HGGs from the CBTN database in order to understand whether PDGFRA inhibitor therapy could be an option for those patients with this mutation.

Mariella Filbin
research
Interests

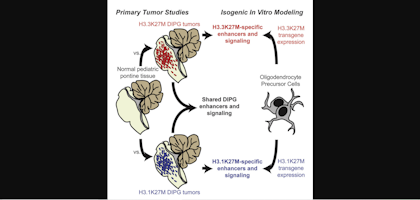
High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
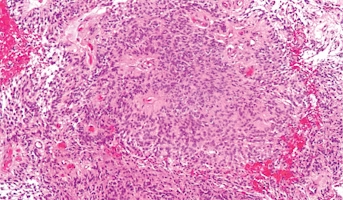
Ependymoma
Ependymomas arise from ependymal cells that line the ventricles and passageways in the brain and the center of the spinal cord. Ependymal cells produce cerebrospinal fluid (CSF). These tumors are classified as supratentorial or infratentorial. In children, most ependymomas are infratentorial tumors
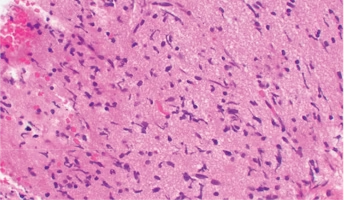
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with