Comprehensive Molecular Analysis of Pediatric Thalamic tumors
Email Principal InvestigatorSupratentorial or Spinal Cord PNET
choroid plexus carcinoma


Javad Nazarian
CBTN Specimen
CBTN Participants
Backer
Smashing Walnuts
About this
Project
Pediatric thalamic tumors are relatively rare cancers and are classified as either low grade gliomas (LGG) or high grade gliomas (HGG). Bilateral thalamic gliomas (BTG) are a particularly rare tumor in this group. Recent molecular studies and WHO classification places thalamic tumors in the category of midline gliomas such as diffuse intrinsic pontine gliomas (DIPGs). This new categorization has been mainly based on discoveries that show histone 3 mutations to drive both of these tumors types. Where recent studies have identified histone mutations associated with DIPGs, more research is required to provide a clear landscape of genomic aberrations associated with thalamic tumors. The goals of this project are to generate the comprehensive molecular profile specific to primary thalamic tumors and to validate genomic abnormalities using liquid biopsy and whole exome sequencing (DNA from blood samples). This project is a comprehensive study of the biology of thalamic glioma and understanding its genetic background. This understanding could lead to more accurate diagnostics and more effective treatment options for patients. The Children's Brain Tumor Network contributed to this project by providing rare plasma samples, tumor tissue, tumor DNA, cerebral spinal fluid and germline DNA.
Ask The
Scientists
What are the goals of this project?
Recent studies have reclassified thalamic tumors as midline gliomas and this project seeks to further analyze genomics of these tumors in an effort to develop targeted therapies and test a new approach to diagnosis these tumors using a liquid biopsy which would be a less invasive diagnostic approach as opposed to surgery.
What is the impact of this project?
This research will expand upon recent insight into thalamic tumors in an effort to develop more accurate diagnostics and targeted treatment options.
Why is the CBTN request important to this project?
The samples needed to carry out this research are rare, which is why the Children’s Brain Tumor Networks contribution to this project is so important.
Specimen Data
The Children's Brain Tumor Network contributed to this project by providing plasma samples, tumor tissue, tumor DNA, cerebral spinal fluid and germline DNA.
Meet The
Team
Heloisa Moser
backing
Foundation and Family Partnerships
related
Histologies
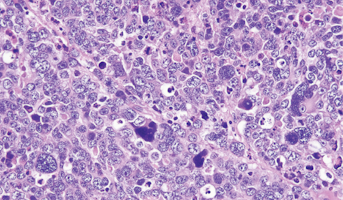
Medulloblastoma
Medulloblastomas comprises the vast majority of pediatric embryonal tumors and by definition arise in the posterior fossa, where they constitute approximately 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% or less of all childhood brain tumors.The clinical feature

High-Grade Glioma
High-grade Gliomas (HGG) or astrocytomas in children nearly always result in a dismal prognosis. Although novel therapeutic approaches are currently in development, preclinical testing has been limited, due to a lack of pediatric-specific HGG preclinical models. These models are needed to help test
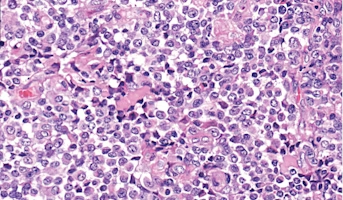
Atypical Teratoid/Rhabdoid Tumor
Central nervous system (CNS) atypical teratoid/rhabdoid tumor (AT/RT) is a very rare, fast-growing tumor of the brain and spinal cord. It usually occurs in children aged three years and younger, although it can occur in older children and adults. About half of these tumors form in the cerebellum or
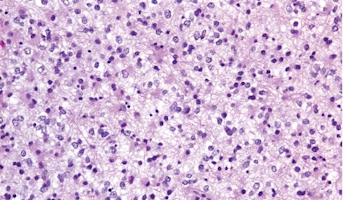
Low-Grade Glioma
Low-Grade Gliomas also called astrocytomas are the most common cancer of the central nervous system in children. They represent a heterogeneous group of tumors that can be discovered anywhere within the brain or spinal cord. Although surgical resection may be curative, up to 20% of children still su
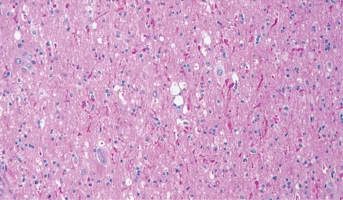
Ganglioglioma
Ganglioglioma presents during childhood and into adulthood. It most commonly arises in the cerebral cortex and is associated with seizures, but also presents in other sites, including the spinal cord.[65,74]The unifying theme for the molecular pathogenesis of ganglioglioma is genomic alterations lea
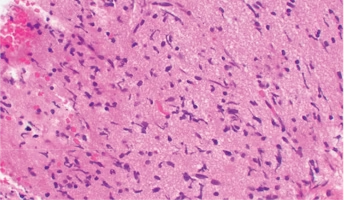
Diffuse Intrinsic Pontine Glioma
A presumptive diagnosis of DIPG based on classic imaging features, in the absence of a histologic diagnosis, has been routinely employed. Increasingly however, histologic confirmation is obtained for both entry into research studies and molecular characterization of the tumor.[5] New approaches with
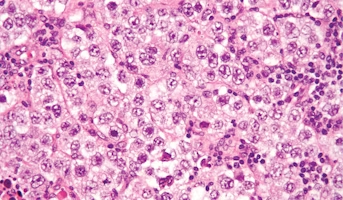
Germinoma
Germinomas are the most common type of CNS germ cell tumor and have a good prognosis.Germ cells are special types of cells that are present as the fetus (unborn baby) develops. These cells usually become sperm in the testicles or unfertilized eggs in the ovaries as the child matures. Most germ cell
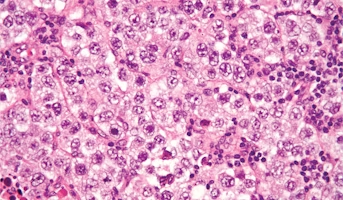
Nongerminomatous Germ Cell Tumors
Primary CNS GCTs are a heterogeneous group of neoplasms that are more common in Japan and other Asian countries than in North America and Europe. In North America, they account for approximately 4% of all primary brain tumors, with a peak incidence from age 10 years to age 19 years and a male predom


